|
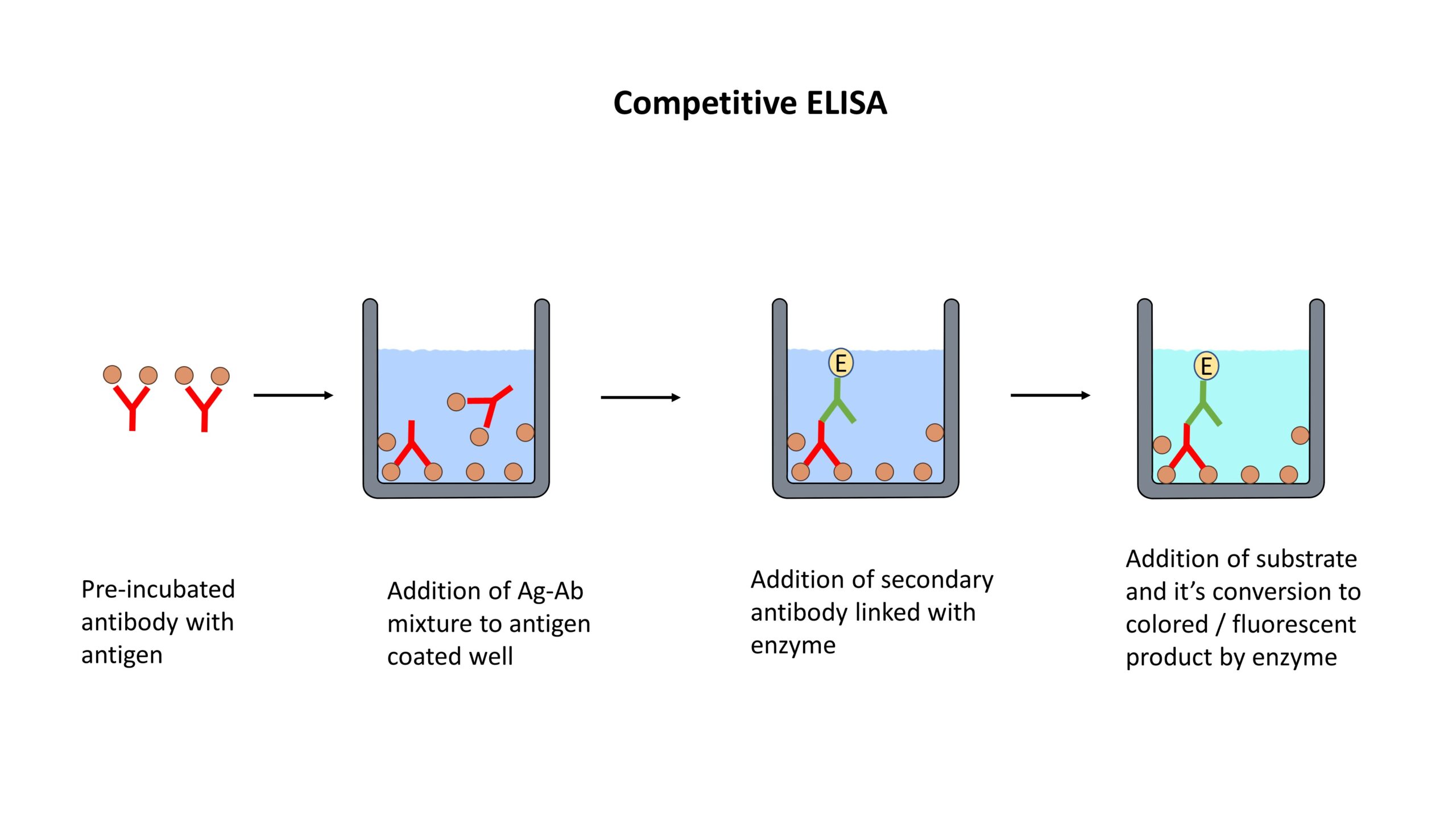
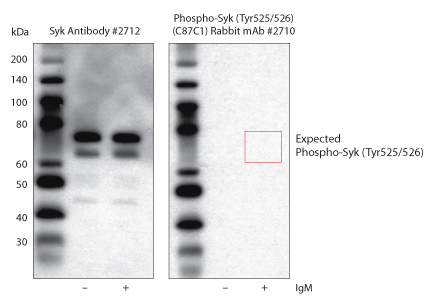
For more details, see: Recommendations for Test Performance and Interpretation from the Second National Conference on Serologic Diagnosis of Lyme Disease. Before CDC will recommend new tests, they must be cleared by the Food and Drug Administration (FDA). New tests may be developed as alternatives to one or both steps of the two-step process. These different methods are based on either colorimetric, chemiluminescence or fluorescence signals for detection. Positive IgM results should be disregarded if the patient has been ill for more than 30 days.ĬDC supports the development of new tests Immunostaining techniques include immunohisto- and immunocytochemistry flow cytometry, enzyme-linked immunosorbent assays (ELISA) and western blots. 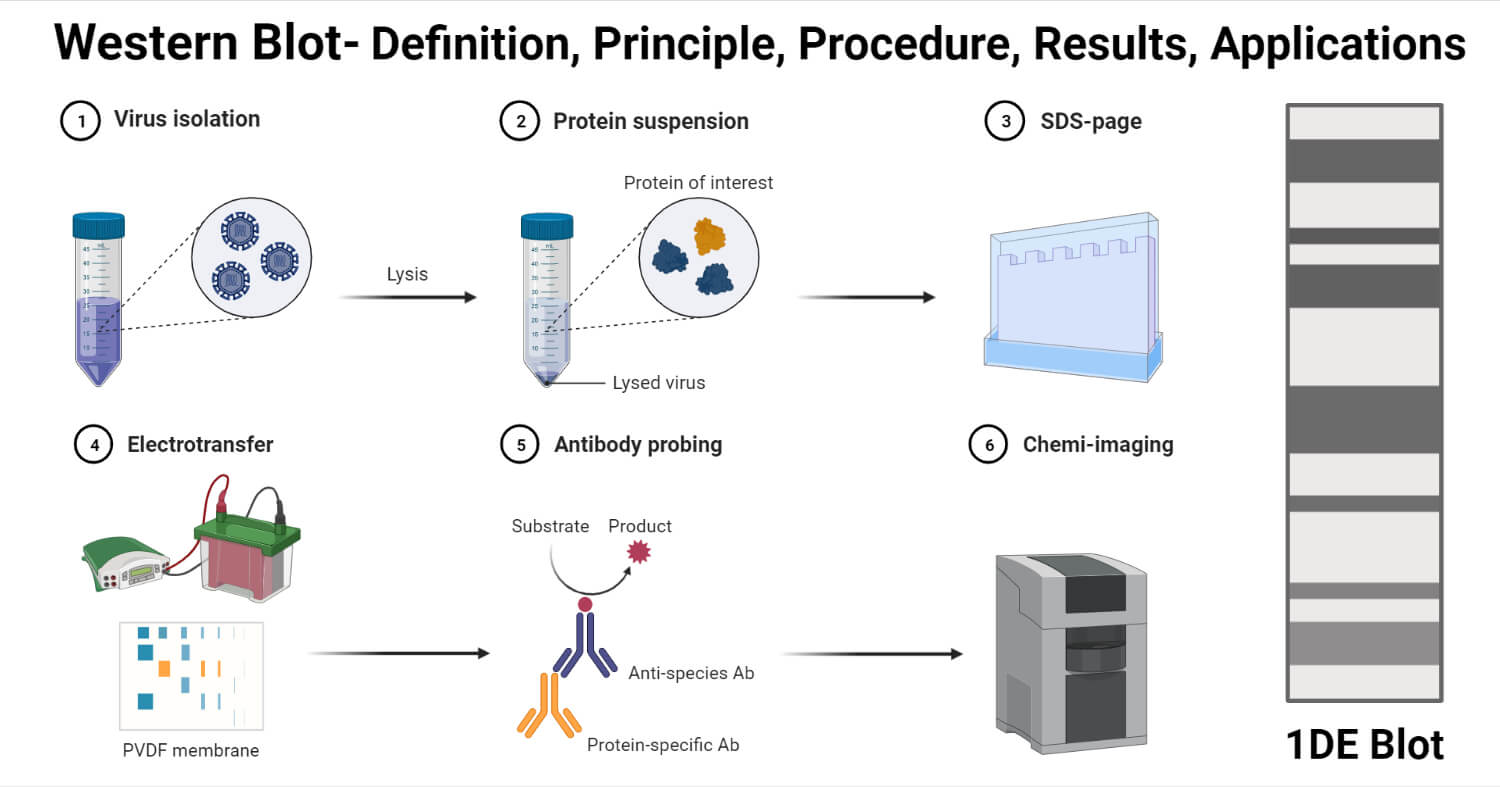
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
